|
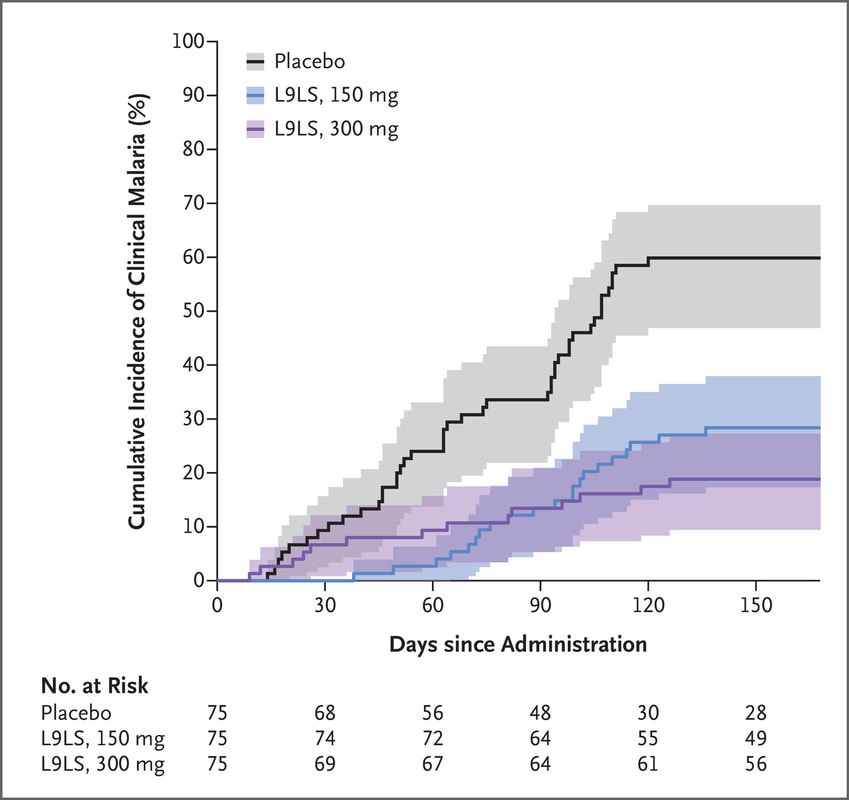
Congratulations Dr. Kassoum Kayentao and the entire L9LS study team for today's publication of the L9LS monoclonal antibody (mAb) clinical trial that was conducted in children 6-10 years old in Mali. The mAb was given subcutaneously, which simplified its administration. The mAb achieved 67-77% efficacy against clinical malaria over six months. Our Malaria Molecular Diagnostic Laboratory performed RT-PCR testing in support of the trial. Looking forward to continuing to work with this great team to help protect children and other vulnerable populations in the fight against malaria.
0 Comments
HELP Unlock the Secrets of Malaria: Join the Murphy Laboratory at the University of Washington!3/5/2024 Are you passionate about making a global impact on health? The Murphy Laboratory at the University of Washington invites talented and ambitious current UW undergraduates to join our groundbreaking research on malaria.
As a member of our dynamic team, you'll have the unique opportunity to work alongside world-renowned scientists and researchers dedicated to understanding the complexities of malaria. Dive into cutting-edge projects aimed at developing new and improved vaccines, exploring innovative diagnostic strategies, and advancing our knowledge of the parasite's biology. Why choose the Murphy Laboratory? 1. Impactful Research: Contribute to projects that have the potential to transform the landscape of malaria treatment and prevention. 2. Collaborative Environment: Work in a supportive and collaborative atmosphere, where your ideas and contributions are valued. 3. State-of-the-Art Facilities: Access cutting-edge laboratory facilities at UW's South Lake Union campus and resources to enhance your research experience. 4. Mentorship: Benefit from mentorship by seasoned researchers, fostering your academic and professional growth. 5. Global Reach: Contribute to a global effort to combat malaria, addressing a critical health challenge on a worldwide scale. Join us in the fight against malaria! For more information, please email Dr. Murphy including '2024 UW Undergrad Research Opportunities' in the subject line. Please provide a CV and outline any prior experiences in laboratories (if any). CongratULATIONS to masters student Nilasha Sen on UW Lab medicine strandjord-clayson award for 2024!2/15/2024 On 2/14/2024, DLMP Masters student Nilasha Sen was announced as the recipient of the Strandjord-Clayson Award in UW Lab Medicine! At the award seminar, Nilasha presented an outstanding talk summarizing the work that she is doing with Pathobiology graduate student Caroline Duncombe for her Master’s thesis under the primary mentorship of Dr. Melanie Shears. Congrats to Nilasha for her achievements and also to Caroline and Melanie for their superb mentorship and generous and skilled approach to training!
More congrats to Nilasha as well for her recent admission to medical school. Excited to see her begin that next chapter in Chicago later this year! We conducted a study with collaborators at Med Biotech in Uganda to assess the natural history of asymptomatic malaria in a malaria-endemic part of the country. The study relied on at-home, participant-collected dried blood spots (DBS) and cost-effective DBS pooling strategies and revealed a high rate of asymptomatic infections and the very dynamic nature of these infections. The study is now published in Lancet Microbe (Assessing the daily natural history of asymptomatic Plasmodium infections in adults and older children in Katakwi, Uganda: a longitudinal cohort study - https://www.sciencedirect.com/science/article/pii/S2666524723002628). More info about the study is also available on the UW Medicine News site (https://newsroom.uw.edu/news-releases/classifying-the-natural-history-of-asymptomatic-malaria). We are grateful to our partners at Med Biotech and to the volunteers for their generous participation in this important study. We hope to use the methods developed and lessons learned from this study to continue our work to understand these low-density infections as the world works toward the ambitious goal of malaria elimination.
Today was my last official day giving microbiology plate rounds at UW Montlake ahead of my clinical move to Seattle Children’s Hospital next week. Since 2011, I have been fortunate to work with the outstanding team in NW177 to care for patients, teach about microbes (especially parasites!) and safeguard our hospital system through the lens of the laboratory. It has been a pleasure every Wednesday to work with this group and to team with partners in Infectious Diseases, Pharmacy, and other departments. Most patients will never know how many caring and knowledgeable people in the microbiology lab helped them get appropriately diagnosed and treated for their illnesses, but many walking out the doors of our hospitals have benefited and even had their lives saved from your service. Thanks for being quality-minded, patient-centered microbiology warriors — you are all amazing!
Cheers to UW Micro, Sean Exciting times as we pursue a highly-effective malaria vaccine in HIGHLY-TRANSLATIONAL animal models10/4/2023 Our team has been working for years to develop a highly effective malaria vaccine that works by inducing a specific kind of T cell immunity in the liver called 'liver resident memory T cells'. These cells are meant to populate the liver and then respond to a future Plasmodium sporozoite infection by more rapidly alerting additional immune cells to completely eliminate any and all parasite-infected cells before they can develop into the disease-causing blood stage. We can protect mice with our vaccines with relative ease, but it has been difficult to obtain consistently good vaccine-induced immune responses in non-human primates (NHPs). The latest improvements to our vaccine approach may have overcome that bottleneck as we have now seen improved responses in our NHPs. Below is a photo of Drs. Felicia Watson and Melanie Shears inspecting a dried interferon-gamma ELISPOT plate for these type of responses. These sort of immunization/challenge experiments take a long time (many months!) and we eagerly await the post-challenge results expected late this year. If successful, next steps for this type of approach could include moving into a human clinical trial in the coming years.
Congrats to Dr. Melanie Shears, our team, and collaborators for publishing our report on our first prime-and-trap vaccination study in non-human primates (NHPs). This study provides critical insight about T cells that reside in the liver of vaccinated animals by showing that prime-and-trap vaccination can induce high levels of antigen-specific cells in the liver, but the response is more variable than in rodent models. We are working on newer prime-and-trap vaccine approaches to improve the reliability of the liver-specific immune response. Check out the manuscript here: https://www.sciencedirect.com/science/article/pii/S0264410X23009131?dgcid=author
Congrats to Dr. Guanyou Lin for his research on DNA and RNA nanoparticles in the UW Department of Materials Science & Engineering. Dr. Lin conducted his PhD thesis research in the laboratory of Dr. Miqin Zhang whose nanoparticle research program spans a variety of uses including drug delivery, diagnostic imaging, and gene therapy. Dr. Lin's PhD resulted in 15 papers! It was a privilege to serve on Dr. Lin's thesis committee and to watch his tremendous research productivity (even when the COVID-19 pandemic arrived mid-PhD!). Congratulations to Dr. Lin!
It's official! Congratulations to Dr. Felicia Watson who graduated today with her PhD in Pathobiology at UW. Felicia's thesis was entitled "Preclinical optimization of a prime-and-trap malaria vaccine". Felicia's malaria vaccine research resulted in two published first authors papers (PMID: 36671393 and
35226868) plus a third groundbreaking study that is currently under review. Today Dr. Watson also gave the student speech on behalf of the Pathobiology PhD graduates (bottom photo) where she highlighted the advantages of UW's Pathobiology Program and the exciting prospects for today's graduating class. We in the malaria field are lucky that Felicia joined our community and are grateful for her commitment to global health! Congrats Dr. Watson! Congratulations to Shruthi Shankar Raman who defended her Masters in Laboratory Medicine today at UW South Lake Union. For her Masters research, Shruthi refined a flow cytometry-based surrogate marker assay for T cell activation in peripheral blood. This assay can now be used in the laboratory for serial sampling to monitor immune responses over time in rodent models of malaria. The Masters in Laboratory Medicine is supported by the Department of Laboratory Medicine and Pathology (link).
|
Lab PostUpdates about lab activities, papers and other insights! Archives
April 2024
Categories |














 RSS Feed
RSS Feed