See below for Malaria / Other pathogens & diseases
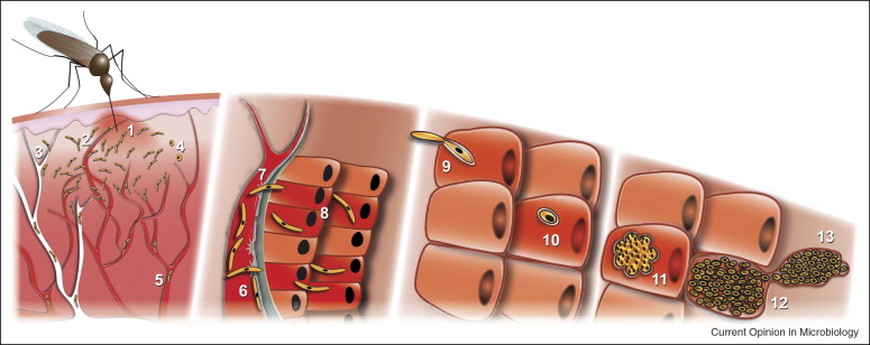
Human Plasmodium infection starts when sporozoite-stage parasites are injected into the dermis by infected female Anopheles mosquitoes as they take a bloodmeal. Within minutes to hours, sporozoites migrate out of the dermis and travel to the liver where they infect hepatocytes and replicate. Liver infection proceeds without any clinical signs or symptoms and produces thousands of asexual merozoites in each infected hepatocyte. For P. falciparum, maturation through the liver takes ~5-7 days and yields ~20,000-30,000 merozoites per infected host cell. Merozoites are released into the blood where they invade red blood cells (RBCs). The parasites go through repeated cycles of invasion and growth in RBCs, leading to clinical symptoms including fevers and chills and potentially progressing to severe anemia, respiratory distress, cerebral malaria, coma and even death without treatment.
We focus our vaccine discovery efforts on the sporozoite and liver stages because there are relatively few parasites at this point in the lifecycle (just 100s to 1000s) compared to the RBC stage. In the RBC there are billions and billions. One milliliter of blood contains about four billion RBCs so with six liters of blood in the body where 1% of the RBCs are infected there's 240 billion RBC-stage parasites to kill - YIKES! Killing off the liver stage parasites can prevent the RBC stage infection...but only if you kill off 100% of the infected cells. You lose if even one infected hepatocyte survives to release its merozoites into the bloodstream because the mechanisms of immunity against liver and blood stages are so different.
Our goal is to make a multi-component subunit vaccine that teaches the immune system to target proteins expressed by the sporozoites AND proteins expressed later in infected hepatocytes. We liken the process of killing all of the infected liver cells to how one might approach a tumor. First, like a surgeon, we utilize strong antibody and T cell responses to sporozoite-expressed proteins as an immunogical 'scalpel' whose job is to significantly cut down on the incoming load of parasites that successfully invade hepatocytes. However, just one parasite is enough to cause RBC infection later on. Therefore, like an oncologist, we next aim to add additional vaccine-induced immune responses to proteins expressed later in the liver stage. These late responses would act like immune 'chemotherapy' to consolidate and kill the last few remaining infected cells. Getting to ZERO is critical because just like in cancer, further development of a single abnormal cell gives rise to the RBC stage and results in failure to achieve sterile protection.
We and other groups worldwide have recently appreciated the critical importance of a particular type of CD8+ T cell called the liver resident memory T cell or Trm cell. Resident memory T cells are probably important in many types of localized infections. In the pre-erythrocytic stage of malaria parasite, Trm cells in the liver are critical for finding and eliminating infected hepatocytes before the red blood cell stage can begin. We are pursuing prime-and-trap strategies aimed at raising the frequency of these specialized cells.
For our immunology/vaccinology studies, we use a variety of tools and methods including:
- T cell screening by IFN-gamma ELISPOT using antigen presenting cells transfected with minigene libraries encoding candidate antigens.
- Immunization of mice using attenuated whole sporozoites (P. yoelii and P. berghei) and using nucleic acid vaccines delivered by gene gun, electroporation, and other methods
- Challenge of mice with sporozoites with endpoints that include In Vivo Imaging of luciferase-expressing parasites as well as Giemsa-stained blood smears and RT-PCR
- Multi-parameter flow cytometry to evaluate antigen-specific immune cell populations in the liver, spleen, and blood
- Tissue-specific vaccination using strategies such as prime-and-trap
- In vitro culture-based experiments to understand parasite biology as it relates to immune responses
- DNA cloning, in vitro transcription, Western blotting, and more...
We focus our vaccine discovery efforts on the sporozoite and liver stages because there are relatively few parasites at this point in the lifecycle (just 100s to 1000s) compared to the RBC stage. In the RBC there are billions and billions. One milliliter of blood contains about four billion RBCs so with six liters of blood in the body where 1% of the RBCs are infected there's 240 billion RBC-stage parasites to kill - YIKES! Killing off the liver stage parasites can prevent the RBC stage infection...but only if you kill off 100% of the infected cells. You lose if even one infected hepatocyte survives to release its merozoites into the bloodstream because the mechanisms of immunity against liver and blood stages are so different.
Our goal is to make a multi-component subunit vaccine that teaches the immune system to target proteins expressed by the sporozoites AND proteins expressed later in infected hepatocytes. We liken the process of killing all of the infected liver cells to how one might approach a tumor. First, like a surgeon, we utilize strong antibody and T cell responses to sporozoite-expressed proteins as an immunogical 'scalpel' whose job is to significantly cut down on the incoming load of parasites that successfully invade hepatocytes. However, just one parasite is enough to cause RBC infection later on. Therefore, like an oncologist, we next aim to add additional vaccine-induced immune responses to proteins expressed later in the liver stage. These late responses would act like immune 'chemotherapy' to consolidate and kill the last few remaining infected cells. Getting to ZERO is critical because just like in cancer, further development of a single abnormal cell gives rise to the RBC stage and results in failure to achieve sterile protection.
We and other groups worldwide have recently appreciated the critical importance of a particular type of CD8+ T cell called the liver resident memory T cell or Trm cell. Resident memory T cells are probably important in many types of localized infections. In the pre-erythrocytic stage of malaria parasite, Trm cells in the liver are critical for finding and eliminating infected hepatocytes before the red blood cell stage can begin. We are pursuing prime-and-trap strategies aimed at raising the frequency of these specialized cells.
For our immunology/vaccinology studies, we use a variety of tools and methods including:
- T cell screening by IFN-gamma ELISPOT using antigen presenting cells transfected with minigene libraries encoding candidate antigens.
- Immunization of mice using attenuated whole sporozoites (P. yoelii and P. berghei) and using nucleic acid vaccines delivered by gene gun, electroporation, and other methods
- Challenge of mice with sporozoites with endpoints that include In Vivo Imaging of luciferase-expressing parasites as well as Giemsa-stained blood smears and RT-PCR
- Multi-parameter flow cytometry to evaluate antigen-specific immune cell populations in the liver, spleen, and blood
- Tissue-specific vaccination using strategies such as prime-and-trap
- In vitro culture-based experiments to understand parasite biology as it relates to immune responses
- DNA cloning, in vitro transcription, Western blotting, and more...
In addition to studies on Plasmodium parasites, our laboratory has also worked on other complex pathogens. We developed DNA vaccines for a USDA-funded effort to fight Theileria parva, an apicomplexan parasite that causes East Coast Fever in cattle. Some of this research was published in 2019 with our collaborator Dr. Lindsay Fry of the USDA/Washington State University. Our laboratory is also broadly interested in vaccines for other pathogens and diseases such as Mycobacterium tuberculosis, hepatitis viruses, and cancers.
Go to...










