New paper out in OFID on clearance of the Plasmodium 18S rRNA biomarker after anti-malaria treatment4/16/2023 Our latest diagnostic paper by first authors Chris Chavtur and Weston Staubus is now published in the Open Forum on Infectious Diseases. The paper, entitled "Plasmodium 18S rRNA biomarker clearance after FDA-approved antimalarial treatment in controlled human malaria infection trials" demonstrates the clearance profile of this diagnostic marker after definitive treatment. The study's findings are based on numerous human challenge studies conducted with our collaborators and will help refine and streamline future challenge study designs.
0 Comments
Our CLIA/CAP-certified MMDL team accomplished a lot in 2022 with RT-PCR samples totaling 10,034 in numerous pre-clinical and clinical studies around the world. The team of Mariko Seilie, Weston Stuabus, Chris Chavtur, and Ming Chang, PhD are committed to high-quality molecular diagnostic testing. We are eager to continue these efforts in 2023 and are grateful for the support from our sponsors and funders. Malaria RT-PCR allows us to detect and quantify infections that are missed by most other assays, and this information can inform vaccine and drug development, malaria epidemiology studies, and elimination efforts.
Congrats to Caroline Duncombe for successfully completing her General Exam in the UW Pathobiology PhD Program! She gave a tremendous seminar describing the great progress she is making toward her thesis project. We are looking forward to seeing her important findings published soon!
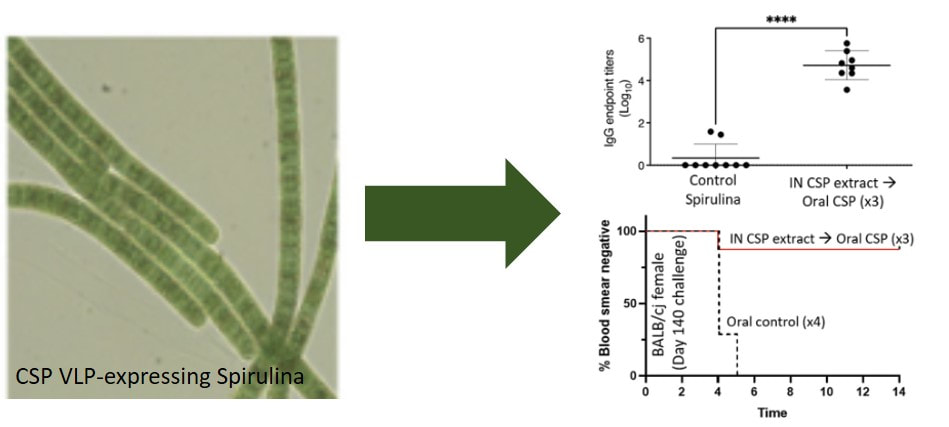
needle-free spirulina-based P. falciparum CSP malaria vaccine study now published in npj vaccines10/4/2022 Our new paper on a Plasmodium falciparum circumsporozoite protein (PfCSP) vaccine composed of PfCSP-bearing VLPs expressed by the edible algae spirulina (scientific name: Arthrospira platensis) is now online in npj Vaccines (LINK). In this Phase 1 STTR-funded study, a combination of intranasal priming and oral boosting achieved high titer anti-PfCSP antibody responses that protected mice against PfCSP-expressing sporozoite challenge up to 140 days post-vaccination. This approach is being further pursued by our team and Lumen Bioscience in a Phase 2 NIH STTR project. If developed, needle-free vaccination approaches have the potential to simplify vaccine administration and improve vaccine uptake.
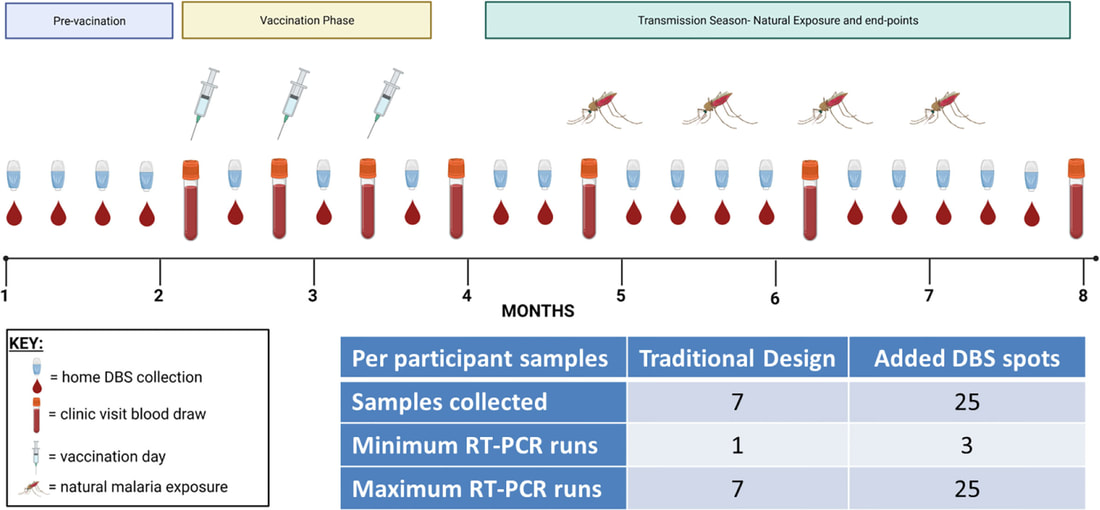
Led by first-author Tonny Owalla, we recently published a forward-looking perspective piece in Frontiers in Immunology's collection 'Puzzle Pieces From Malaria Vaccine Clinical Trials' entitled "Rethinking detection of pre-existing and intervening Plasmodium infections in malaria clinical trials". The paper, which is based on lessons learned from an epidemiology study conducted by Mr. Owalla and our own recent PhD graduate Dr. Dianna Hergott, argues that more frequent and more intensive molecular diagnostic testing is needed before, during, and after malaria vaccination to fully assess the role of underlying P. falciparum and non-P. falciparum infections in sub-optimal vaccine responses (LINK). This approach may sound expensive, but the critical data can be cost-effectively obtained using at-home dried blood spot (DBS) collection and pooled DBS testing as a part of trial design, as we recently demonstrated in Dr. Hergott's feasibility study publication (LINK). More to come when the full data from that study is published in the coming months!
Non-human primates (NHPs) are an extremely important animal model for malaria vaccine development. In this study, purified, vialed, cryopreserved Plasmodium knowlesi sporozoites (PkSPZ) were produced by Sanaria, Inc. and tested in studies by Sanaria, NIH, and UW to show that the PkSPZ product resulted in reliable, reproducible infections in two macaque species. Because PkSPZ are cryopreserved, the PkSPZ approach provides more experimental flexibility in planning and scheduling than when infected mosquitoes are used to inoculate the parasite. This NHP/PkSPZ model provides a rigorous experimental system that can be used to study anti-malarial immune responses including those that are inaccessible in humans. The study was led by Dr. Sumana Chakravarty at Sanaria. UW Acting Instructor Dr. Melanie Shears led the UW's pigtailed macaque NHP research in this study with support from the Washington National Primate Research Center P51 award (link).
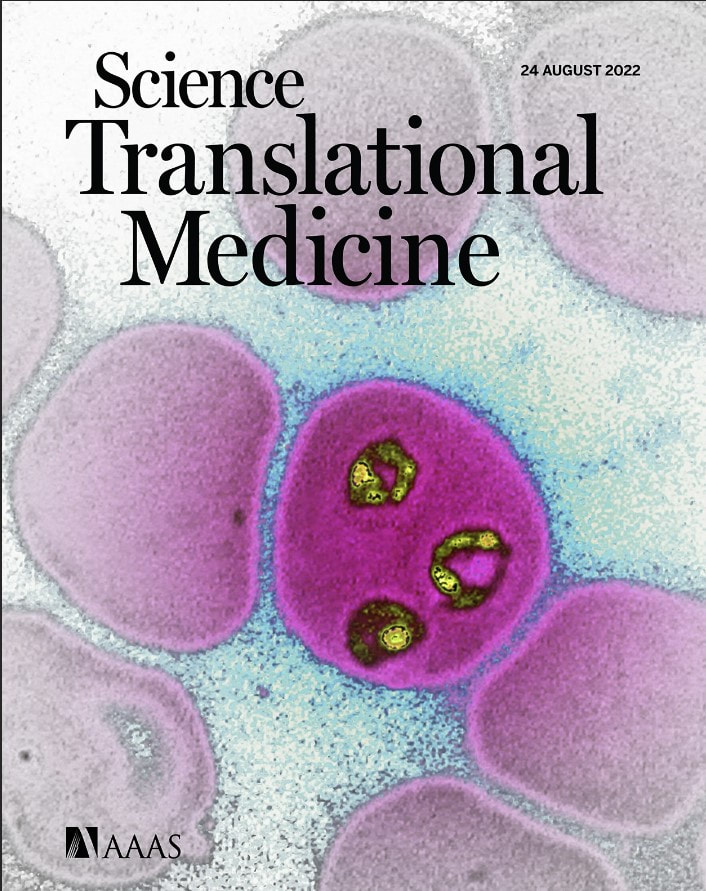
The PfGAP3KO genetically-attenuated parasite vaccine clinical trial is now published in Science Translational Medicine. In this study, participants were immunized three or five times with ~200 mosquito bites per dose containing the PfGAP3KO vaccine. Later, the participants underwent controlled human malaria infection and half of vaccinated participants were protected. The study shows the promise of GAP vaccines and paves the way for additional research to translate these findings to an even more efficacious and deployable vaccine. Thanks to the many partners involved in this study and to the volunteers for their generous participation in this study.
UW Summer Science Program (SSP) student Aida Chan wowed our lab team on August 16 with a stellar presentation summarizing her molecular diagnostic experiments and plans for her remaining immunology-focused experiments. Aida's time in the lab was made possible through the UW SSP and by generous time commitments from Murphy Lab team members Chris Chavtur, Anya Kalata, Weston Staubus, Dr. Rebekah Reynolds, and Mariko Seilie. Thanks to Aida and the team for your efforts all summer long! Thanks also to the UW SSP for sponsoring such a valuable experiential learning program.
Congratulations to Shruthi Shankar Raman for being awarded a Strandjord-Clayson Traineeship Award from the Department of Laboratory Medicine and Pathology for the 2022-2023 academic year. This award recognizes Shruthi's academic excellence and commitment to her Master's training program. Her work in the laboratory is focused on the behavior of CD8+ T cells in the setting of malaria sporozoite vaccination and challenge. We are looking forward to seeing the data emerge from her projects. Thanks to DLMP for this generous support!
July 29, 2022 was a wonderful evening to celebrate the progress we've made on malaria in the past year. The lab team, families, significant others, and friends gathered at the Murphy's house for toasts, ping pong, dinner, and lots of camaraderie. Congrats to Dr. Dianna Hergott for completing her PhD this year and to PhD candidates Felicia Watson and Caroline Duncombe for obtaining fellowship funding support. Congrats also to faculty member Dr. Melanie Shears for obtaining her first fundable score on an NIH R21 grant! Cheers to the MMDL team of Mariko Seilie, Chris Chavtur, Weston Staubus, and Dr. Ming Chang for a successful CAP inspection and for tremendous progress on numerous clinical trial projects including a local CHMI study, an important clinical trial in sub-Saharan Africa, and an ongoing FDA project. Congrats and thanks to our vaccine production team led by Anya Kalata who has been busy at work producing high-quality DNA vaccines to support our mouse and non-human primate studies. Dr. Rebekah Reynolds was hailed for her leadership of our U01 project, which just had its first major publication in Nature Communications with Dr. Hai-Quan Mao's laboratory at Johns Hopkins. Dr. Reynolds and Sam Mak are also making amazing progress with the UW-based insectary that we share with Dr. Marion Pepper's nearby UW laboratory. Finally, thanks to our newer lab members Dr. Naveen Yadav, Dr. Ken Boey, Shruthi Shankar Raman, Ethan Conrad, and Emily Scott who are all making great contributions to our team. Finally, thanks to Dr. Reynolds as well for teaching us how to sabre champagne bottles in celebration of all of these achievements!
|
Lab PostUpdates about lab activities, papers and other insights! Archives
March 2024
Categories |




 RSS Feed
RSS Feed